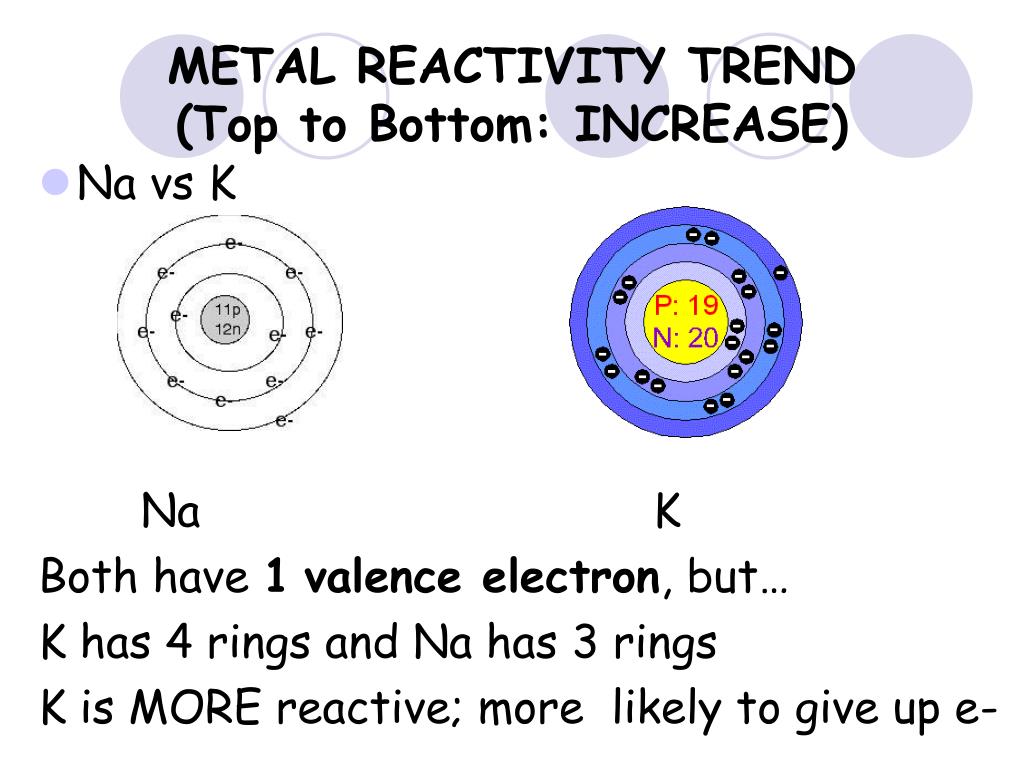
Details and literature references are in the main text. The ensuing dysfunctional astrocyte reactivity exhibits gain- or loss-of-functions that can contribute to further tissue pathology in a vicious cycle. Such disease states can in the absence of astrocyte reactivity, cause or contribute to tissue dysfunctions that in turn lead to production of astrocyte reactivity triggers.
Genetic mutations and polymorphisms can lead to cell-autonomous dysfunctions in astrocytes that lead to non-reactive disease states. Chronic astrocyte reactivity can lead to loss- or gain-of-functions resulting in disease states with dysfunctional reactivity that can exacerbate tissue pathologies and worsen disorder outcome. Astrocyte reactivity can become chronic if triggers persist. Non-proliferative astrocyte reactivity can resolve over time if acute triggers recede. Non-proliferative reactive astrocytes maintain but modify their interactions with surrounding cells in preserved tissue architecture. Non-proliferative astrocyte reactivity can exhibit diverse states with different changes in gene expression and functions that are context dependent as determined by astrocyte starting conditions and incoming reactivity triggers. Newly proliferated astrocytes (red nuclei) form limitans borders that permanently separate and corral areas of damage, inflammation and non-neural scar tissue from adjacent viable neural tissue. The overall trend, for the reactivity of Group 2 metals with water. by traumatic or ischemic cell degeneration, blood-brain barrier leak, infection or autoimmune leukocytic inflammation. Exam tip: You will only need to consider the trends, properties and reactions of the. Proliferative astrocyte reactivity occurs in response to tissue damage caused e.g. Different forms of astrocyte reactivity are triggered by diverse non-cell-autonomous signals emanating from diverse CNS insults. Diverse starting conditions can influence subsequent reactivity responses. Trends of chemical reactivity in Periodic Table The systematic arrangement of elements in a periodic table discloses certain periodic trends in the properties of elements. In healthy tissue, astrocytes exhibit regional and local heterogeneity in gene expression and function. A working model and conceptual framework is presented for characterizing the diversity of astrocyte reactivity.Ĭopyright © 2020 The Author. This review discusses the goal of differentiating reactive astrocyte subtypes and states based on composite pictures of molecular expression, cell morphology, cellular interactions, proliferative state, normal functions, and disease-induced dysfunctions. Nevertheless, concepts about what astrocyte reactivity comprises and what it does are incomplete and sometimes controversial. Long regarded as passive and homogeneous, astrocyte reactivity is being revealed as a heterogeneous and functionally powerful component of mammalian CNS innate immunity. In addition to playing essential roles in healthy tissue, astrocytes exhibit an evolutionarily ancient response to all CNS insults, referred to as astrocyte reactivity. The reason, once more, is that the polarising power of the M 2+ decreases as ionic radius increases.Astrocytes are neural parenchymal cells that ubiquitously tile the central nervous system (CNS). The thermal stability of the nitrates follows the same trend as that of the carbonates, with thermal stability increasing with proton number. This means that beryllium carbonate decomposes at a lower temperature to the rest of the group. The greater the distortion caused by the polarising ion the less stable the compound is to heat. In other words, the greater an elements tendency to gain electrons, the more reactive the element is. repeat step 1 with two more columns of wells, using potassium bromide solution, KBr (aq), then potassium iodide solution, KI (aq) use a dropping pipette to add a few drops of chlorine solution, Cl 2 (aq), to. Hence polarising ability of the M 2+ ion decreases down the group. use a dropping pipette to add a few drops of potassium chloride solution, KCl (aq), to a column of three wells in a spotting tile. The larger the anion the easier the distortion, as seen with the carbonate ion. Small highly charged positive ions distort the electron cloud of the anion. This trend is explained in terms of the Group II metal ions ability to polarise the anion, the carbonate ion. Beryllium carbonate decomposing most easily. The thermal stability of Group II carbonates increases as we descend the group.
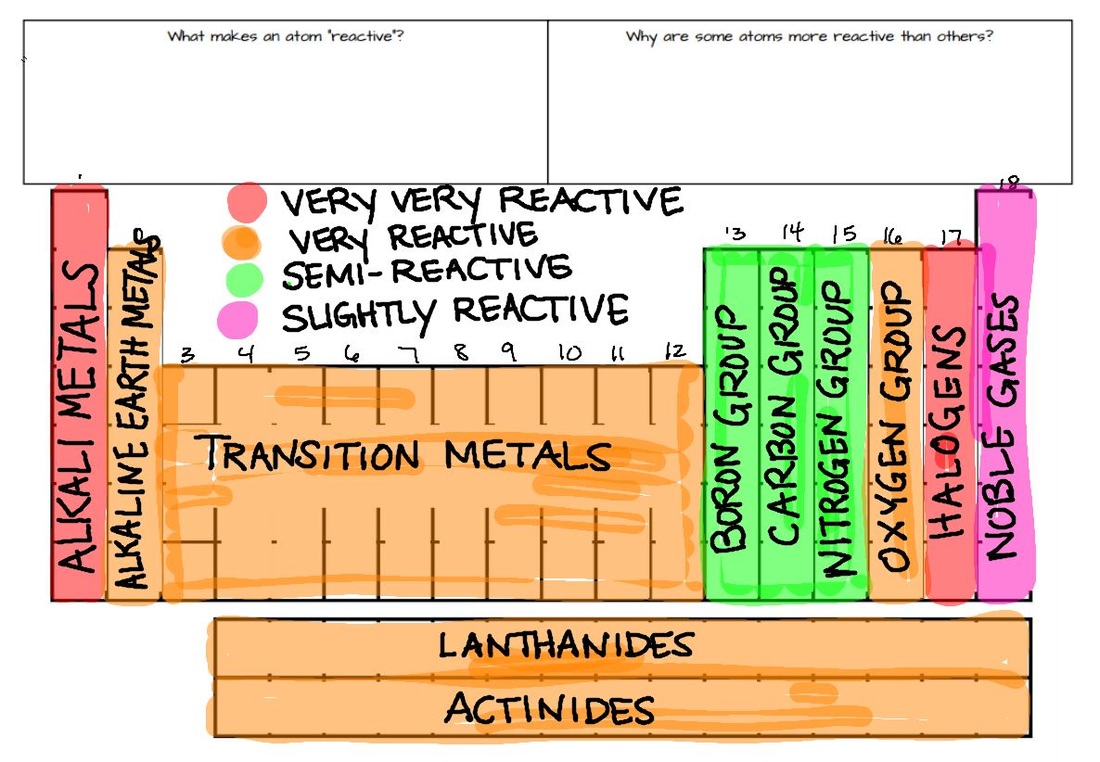
Thermal stability of carbonates and nitrates Carbonates Periodic trends are specific patterns in chemical element properties revealed by the periodic table of elements.
